|
The presence of a lone pair can affect the molecule's reactivity and chemical behavior.The seesaw structure allows for efficient bonding angles and effective positioning of atoms around the central atom.Each fluorine atom contains three lone pairs. Sulfur also possesses one lone pair, giving the molecule its seesaw shape. In SF4, sulfur acts as the central atom attached to four fluorine atoms. Sulfur tetrafluoride (SF4) is an example of a compound with a seesaw molecular structure. Alternatively, the seesaw structure can also be represented with the lone pair opposite to the other atoms. This arrangement gives the molecule a seesaw-like shape. The seesaw structure is another important molecular geometry that involves a central atom attached to four other atoms with one lone pair. The trigonal bipyramidal structure can be challenging to Visualize and understand without proper knowledge of molecular geometry.The trigonal bipyramidal structure allows for efficient placement of five atoms around a central atom, maximizing bonding potential.The total number of valence electrons in this structure is 40. Each chlorine atom also contains three lone pairs. In this structure, phosphorus acts as the central atom, with two chlorine atoms in the z-direction and three chlorine atoms in the xy-plane. These bond angles play a significant role in determining the overall shape and properties of the molecule.Įxample of Trigonal Bipyramidal Structure: PCl5Īn example of a compound with a trigonal bipyramidal structure is phosphorus pentachloride (PCl5). The angle between the atoms on the top and bottom is 180 degrees, while the angle between the three atoms in the middle is 120 degrees. In the trigonal bipyramidal structure, there are three important bond angles to consider. The other three atoms surrounding the central atom have a trigonal planar shape, with a bond angle of 120 degrees between them.īond Angles in Trigonal Bipyramidal Structure There is one atom on top and one on the bottom, with the bond angle between them being 180 degrees. The trigonal bipyramidal structure is a molecular geometry where a central atom is attached to five other atoms. The arrangement of atoms around a central atom determines the molecular geometry and is crucial in determining the chemical properties and behavior of substances. Molecular geometry is a vital concept in chemistry that helps us understand the Shape and arrangement of molecules. 
Example of Square Pyramidal Structure: IF5ħ.1. Example of Trigonal Bipyramidal Structure: PCl5ĥ.2. Bond Angles in Trigonal Bipyramidal StructureĢ.2. So, that bends the axial fluorines together a bit.Unlocking the Secrets of Molecular Geometry with VSEPR TheoryĢ.1.  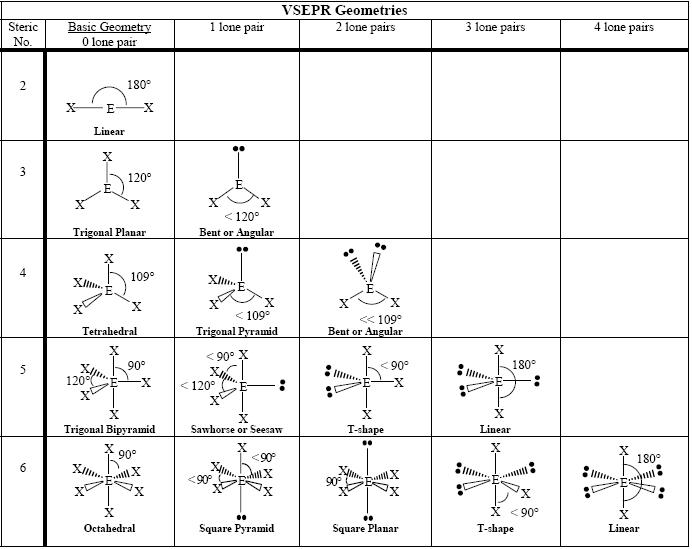
Note though that the structure is distorted a bit due to the repulsive forces of the lone pair of electrons you see (not bonded). With one lone pair of valence electrons, you get a seesaw molecular geometry. Therefore, you can put 6x4 on each fluorine, 2x4 to account for four single bonds, and 2 for the last 2 valence electrons available.Īs a result, you have 5 electron groups, so the electron geometry would be trigonal bipyramidal. You can put sulfur in the middle because fluorine tends to make single bonds. Within the context of VSEPR theory, you can count electrons to determine the electron geometry ("parent" geometry).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
